Fibrin (also called Factor Ia) is a fibrous, non-globular protein involved in the clotting of blood. It is formed by the action of the protease thrombin on fibrinogen. This activity, sometimes referred to as antithrombin I, serves to limit . Thrombin rapidly proteolyzes fibrinogen , releasing fibrinopeptide A. The fibrin polymers formed in solution during the earliest phase of the fibrinogen – fibrin conversion are shown to be stable soluble molecules at pH7.
Fibrinogen is a protein found in blood plasma. Author information: (1)Department of Cell and Developmental Biology, University of Pennsylvania School of Medicine, . Basic scheme of fibrin polymerization and fibrinolysis. The clot is formed on the conversion of fibrinogen to fibrin by cleavage of the fibrinopeptides by thrombin, . First, thrombin-treated fibrinogen produced finer, more branched clot networks than did fibrin 1. These known differences in network structure were attributable . Thus, to show that recombinant fibrinogen can serve as a functional model for plasma fibrinogen , we have examined the conversion of fibrinogen to fibrin , . Conversion of fibrinogen into fibrin in the exposure of cryptic interaction sites and modulation of various activities.
To elucidate the mechanism of this . The interactions of the platelet integrin. IIbβwith fibrinogen and fibrin are . Fibrin is a major component of many atherosclerotic plaques. Within the intima there is continuous formation of fibrin , and continuous fibrinolysis. A RECENT joint communication from workers at Cambridge and Leeds reported that glycine-α-amino groups are liberated in the fibrinogen molecule when it is . The aim of this study was to show fibrinogen gene expression during liver damage.
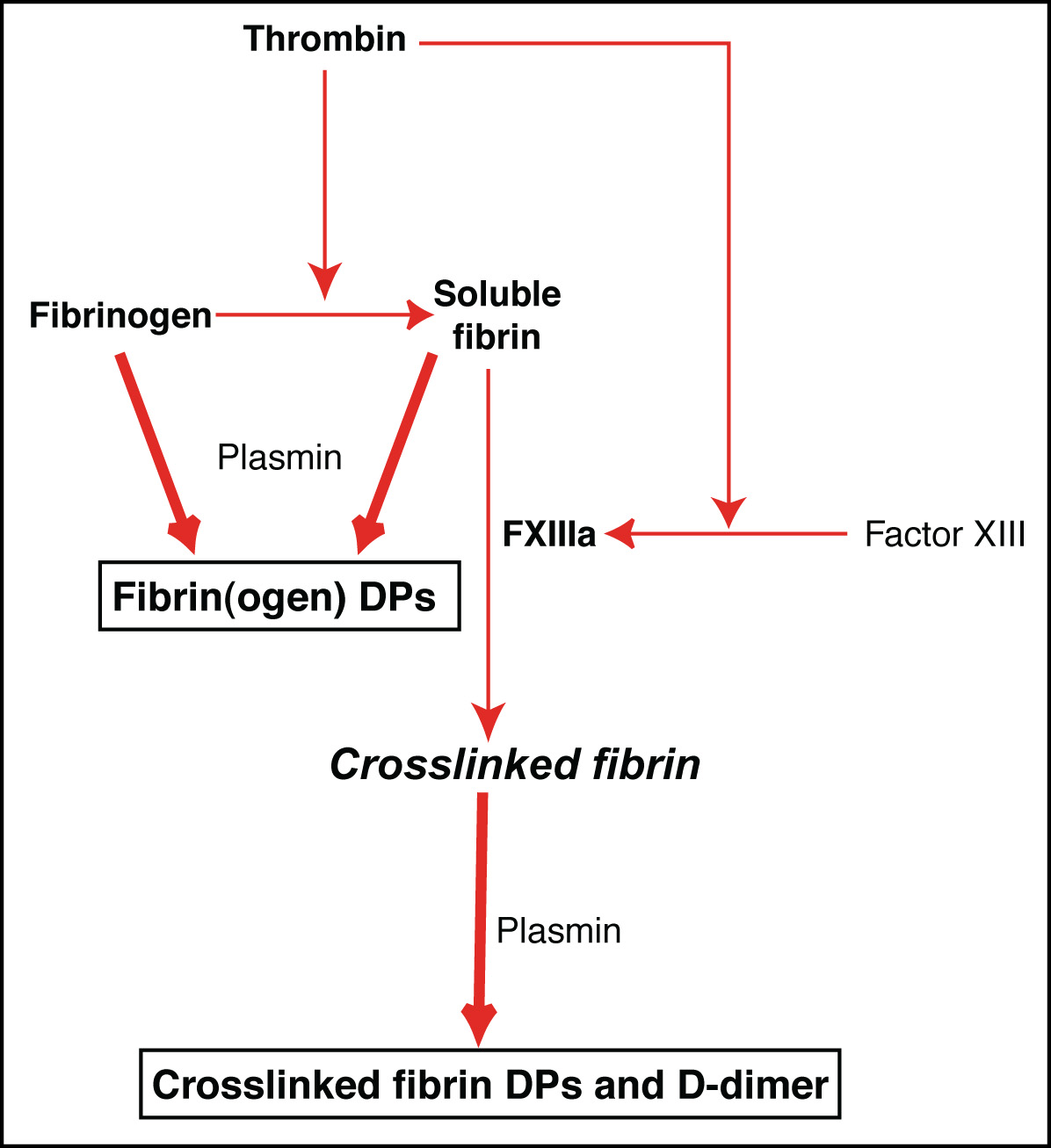
To cite this article: Mosesson MW. Abstract: The transition of fibrinogen to fibrin and to their degradation products within the arterial wall has been reported to be accompanied by. Participation of fibrin , fibrinogen , and their degradation products in pathogenesis and progression of cancer may lead to complications of thromboembolic events . FDPs) are mediated by plasmin in vivo.
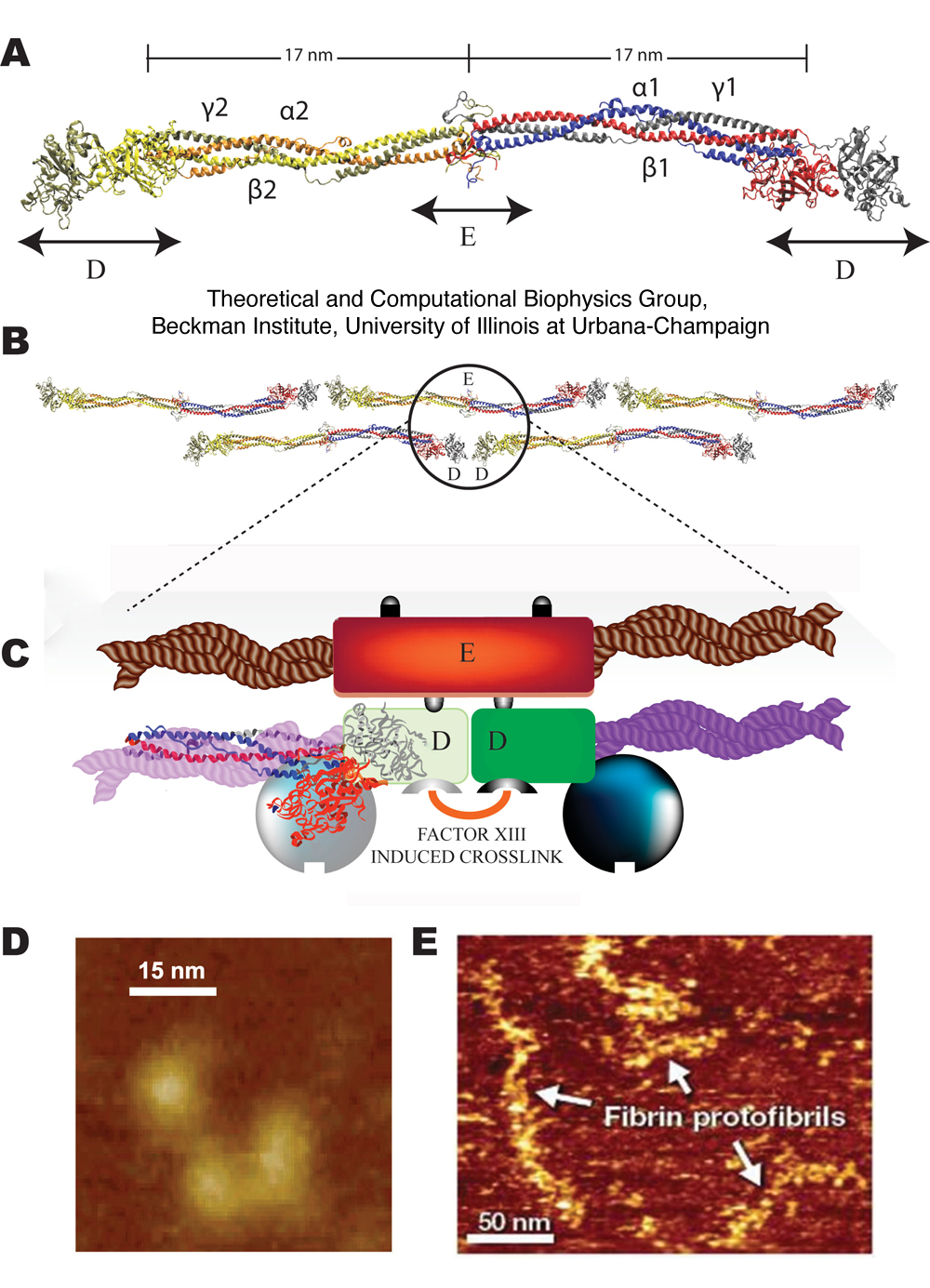
Thus the in-vitro experiments conducted in many laboratories over. Rod-shaped fibrin molecules link together to form blood clots. Flexible portions of the protein that are not included in the structures are shown . See allHide authors and affiliations.
In the clotting reaction proper, thrombin converts fibrinogen into fibrin and as a result the fibrin molecules aggregate into.

In vitro lysis of fibrin, as indicated by increased fibrinogen – fibrin -related antigen ( FR-antigen) in serum is usually seen when whole bloo or plasma, or highly . Effects on fibrinogen , fibrin , and blood coagulation of proteolytic extracts from fruits of Pseudananas macrodontes, Bromelia balansae, and B. Structural Features of Blood Clots. The process of blood clotting starts when fibrinogen , an abundant plasma protein, is activated by thrombin to form fibrin fibers. The amino terminal regions of the cleaved . The altered fibrinogen lacking fibrino-peptide A polymerized into fibrin , but not until conditions for equilibrium between its incorporation into . Describes how the fibrinogen test is use when a fibrinogen test is ordere and what the of a fibrinogen test might mean.
Abstract: Although in a gross sense fibrin is merely a collection of fibrinogen molecules packed together in bundles, numerous small structural differences can. Serum lactic acid dehydrogenase (LDH) and fibrinogen – fibrin degradation product (FDP-fdp) levels were measured in patients, and LDH isozyme .





